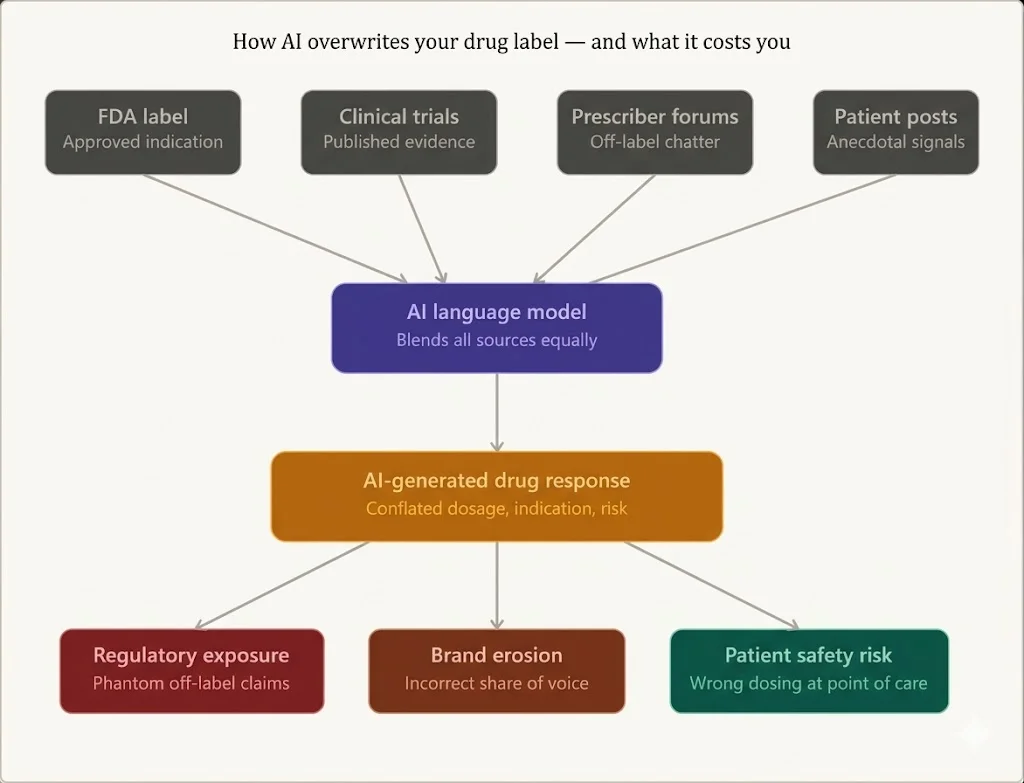
AI Doesn’t Follow Your Drug Label
Large language models don’t rely on a single source of truth. They synthesize answers from a wide range of inputs—often blending regulated and unregulated data.
- 📄 Clinical trials: Emerging and exploratory uses
- 🧑⚕️ Prescriber forums: Real-world off-label practices
- 💬 Patient discussions: Anecdotal outcomes and side effects
- 📚 Publications & abstracts: Early-stage evidence
- 🌐 Web content: Variable quality, often unverified
AI systems are designed to pull and combine information across sources to generate responses, rather than strictly adhering to a single controlled document.
If you’re not monitoring this, you’re not controlling your drug’s narrative.